If you are searching for high-growth Laboratories Indore job opportunities, the latest hiring announcement from Ipca Laboratories Limited is an excellent opportunity. The company is recruiting skilled professionals across API Production, Engineering, Stores, and EHS departments at its Indore facility.
This recruitment drive is ideal for candidates with experience in regulated pharmaceutical environments such as USFDA, WHO-GMP, and MHRA-approved plants.
About Ipca Laboratories Limited
Ipca Laboratories Limited is a globally recognized pharmaceutical company with:
-
Presence in over 120+ countries
-
Strong expertise in Active Pharmaceutical Ingredients (API) and formulations
-
Regulatory approvals from USFDA, WHO-GMP, MHRA
-
Commitment to innovation, quality, and global healthcare
The company’s Indore (Pologround) plant is known for its world-class API manufacturing infrastructure, offering excellent career growth for pharma professionals.
Job Overview – Laboratories Indore Hiring at Ipca
| Category | Details |
|---|---|
| Company | Ipca Laboratories Limited |
| Location | Pologround, Indore |
| Industry | Pharmaceutical (API Manufacturing) |
| Departments | Engineering, Production, Stores, EHS |
| Experience | 2–8+ Years |
| Qualification | B.Sc / M.Sc / ITI / UG / PG |
Available Job Positions
1. Engineering – Electrical & Instrumentation (API)
-
Designation: Assistant Manager / Senior Executive
-
Experience: Minimum 8 Years
Key Responsibilities:
-
Managing transformers, panels, and utilities (DG sets)
-
Calibration of instruments and sensors
-
Troubleshooting electrical faults
-
Ensuring safety and reliability of systems
-
Leading engineering teams
2. Engineering Stores
-
Designation: Officer
-
Experience: Minimum 4 Years
Key Responsibilities:
-
Managing inventory of engineering spares and consumables
-
Stock verification and audit handling
-
Maintaining accurate store records
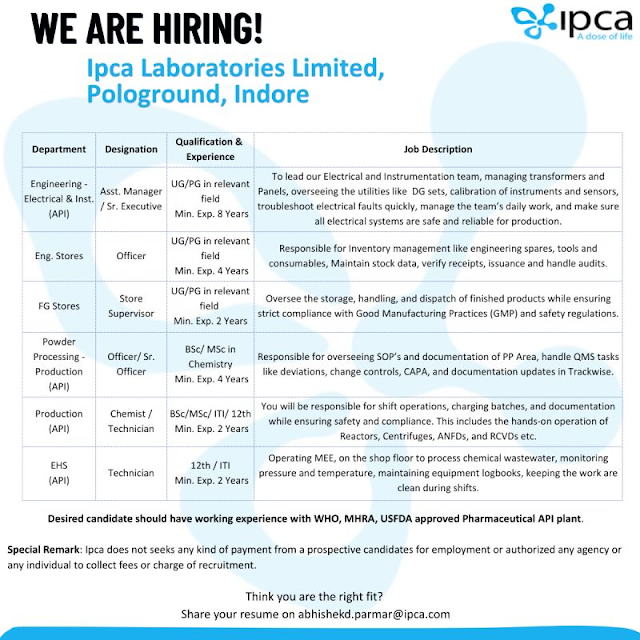
3. Finished Goods (FG) Stores
-
Designation: Store Supervisor
-
Experience: Minimum 2 Years
Key Responsibilities:
-
Handling storage and dispatch of finished products
-
Ensuring compliance with GMP guidelines
-
Maintaining safety and documentation standards
4. Powder Processing – Production (API)
-
Designation: Officer / Senior Officer
-
Experience: Minimum 4 Years
-
Qualification: B.Sc / M.Sc Chemistry
Key Responsibilities:
-
Managing SOPs and documentation
-
Handling QMS activities:
-
Deviations
-
Change Control
-
CAPA
-
-
Updating systems like Trackwise
5. Production (API)
-
Designation: Chemist / Technician
-
Experience: Minimum 2 Years
Key Responsibilities:
-
Operating reactors, centrifuges, ANFDs, and RCVDs
-
Handling batch charging and shift operations
-
Maintaining production documentation
-
Ensuring safety compliance
6. EHS (Environment, Health & Safety)
-
Designation: Technician
-
Experience: Minimum 2 Years
Key Responsibilities:
-
Operating MEE systems for wastewater treatment
-
Monitoring pressure and temperature
-
Maintaining equipment logs
-
Ensuring clean and safe work environment
Who Can Apply?
The Laboratories Indore hiring drive is open to:
-
Candidates with experience in regulated API plants
-
Professionals from WHO, USFDA, or MHRA approved facilities
-
Candidates meeting role-specific qualifications and experience
Selection Process
The hiring process includes:
-
Resume screening
-
Technical evaluation
-
HR interview
-
Final selection
Candidates are assessed based on technical expertise, GMP knowledge, and practical experience.
Why Choose Ipca Laboratories?
Working at Ipca Laboratories Limited offers:
-
Exposure to global pharmaceutical markets
-
Work experience in regulated API manufacturing plants
-
Strong focus on quality, compliance, and innovation
-
Career growth in a globally recognized organization
-
Learning opportunities with advanced pharmaceutical technologies
Ipca’s “A Dose of Life” philosophy reflects its commitment to improving healthcare worldwide.
How to Apply
Interested candidates can apply by sending their updated CV:
📧 Email: abhishekd.parmar@ipca.com
📌 Subject Example: Application for API Production – Chemist Role
Industry Insights – Growth of API Manufacturing in India
India is a global hub for API manufacturing, driven by:
-
Increasing demand for generic medicines worldwide
-
Government initiatives supporting pharmaceutical exports
-
Expansion of regulated manufacturing facilities
Companies like Ipca Laboratories are at the forefront of this growth, creating strong demand for skilled professionals in production, engineering, and quality systems.
Authoritative Sources
For industry standards and compliance guidelines:
-
Central Drugs Standard Control Organization (CDSCO)
-
World Health Organization – GMP Guidelines
-
Medicines and Healthcare products Regulatory Agency
-
United States Food and Drug Administration
These authorities define the regulatory standards followed by companies like Ipca.
FAQs – Laboratories Indore Recruitment 2026
1. Is regulatory plant experience mandatory?
Yes, candidates with experience in WHO, USFDA, or MHRA approved plants are preferred.
2. What is the job location?
All roles are based at Ipca Laboratories, Pologround, Indore.
3. Who can apply for Production Technician roles?
Candidates with B.Sc, M.Sc, ITI, or even 12th qualification with relevant experience can apply.
4. How can I apply?
You can apply by emailing your CV to abhishekd.parmar@ipca.com.
5. Are there any recruitment fees?
No, Ipca Laboratories does not charge any fees during the hiring process.

