Ipca Laboratories Limited is a globally recognized pharmaceutical company known for its strong presence in APIs, formulations, and intermediates. With multiple regulatory approvals including USFDA, MHRA, and WHO-GMP, the company operates advanced manufacturing facilities across India.
Its Indore (Pologround) facility is a major hub for API manufacturing, producing high-quality medicines in therapeutic areas like anti-malarials, cardiovascular, anti-diabetics, and pain management.
Ipca Laboratories Walk-in Interview 2026 – Overview
The latest Ipca Laboratories Walk-in Interview 2026 offers a golden opportunity for experienced professionals in API manufacturing. The company is conducting a walk-in drive for multiple roles in Production, Engineering, Stores, and IT departments.
This hiring drive is ideal for candidates with hands-on experience in regulated pharmaceutical environments.
Walk-in Interview Details
- Date: 29th March 2026 (Sunday)
- Time: 11:00 AM – 3:00 PM
- Venue: Pologround
- Company: Ipca Laboratories Limited
Job Details
- Experience: 2–8+ Years (Pharma/API mandatory)
- Qualification: B.Sc / M.Sc / ITI / 12th / UG/PG / B.E (IT/CS)
- Work Type: On-site (Shift duties applicable)
Open Positions
🔧 Engineering Roles
- Engineer – Electrical & Instrumentation (8+ years)
- Officer – Engineering Stores (4+ years)
Production Roles
- Officer / Sr. Officer – Powder Processing (API)
- Chemist / Technician – Production
Stores Roles
- Store Supervisor – Finished Goods
IT Roles
- Officer – IT (Infrastructure / Network / Compliance)
Required Skills
- Experience in USFDA/MHRA/WHO-GMP approved API plants
- Knowledge of GMP, SOPs, QMS (Trackwise, CAPA, deviations)
- Hands-on experience with API equipment (Reactors, ANFD, RCVD)
- Engineering utilities, calibration, and troubleshooting skills
- IT infrastructure, server/network monitoring expertise
Key Responsibilities
- Manage production operations and batch processing
- Handle engineering utilities, calibration, and maintenance
- Maintain GMP compliance in stores and dispatch
- Execute documentation and QMS processes
- Support IT systems, infrastructure, and compliance
- Ensure safety, quality, and operational efficiency
Benefits & Perks
- 🌍 Work in a globally compliant API manufacturing company
- 📈 Strong career growth opportunities
- 🧪 Exposure to USFDA/MHRA/WHO-GMP standards
- 💰 Competitive salary (no bar for right candidate)
- 📚 Continuous learning and regulatory training
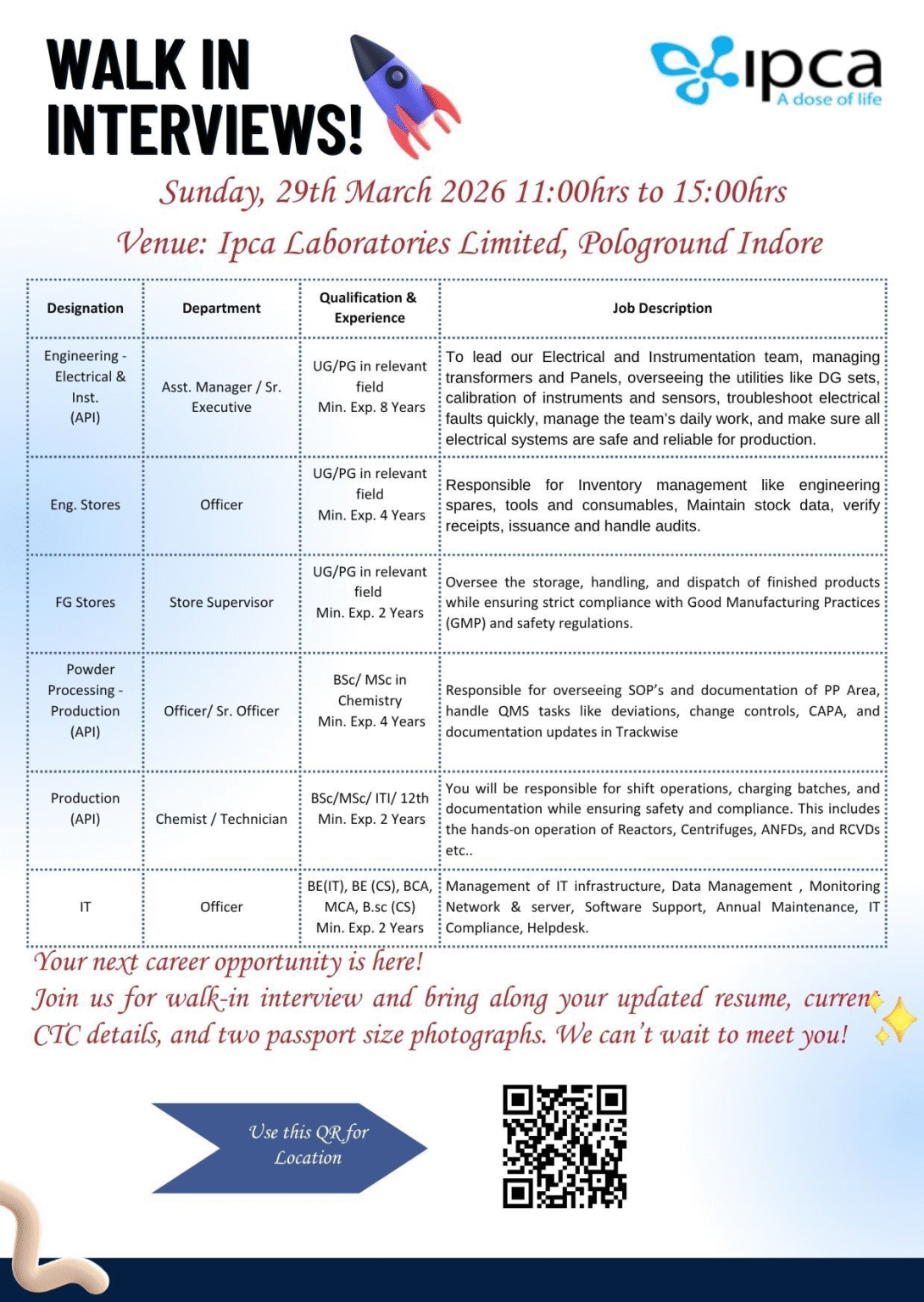
How to Apply
- Attend the walk-in interview on 29th March 2026
- Carry:
- Updated resume
- Current CTC details
- Two passport-size photographs
- Candidates unable to attend can share their CV via official HR email (check original posting)
Industry Insight
API (Active Pharmaceutical Ingredient) manufacturing is a critical segment of the pharmaceutical industry, requiring strict adherence to global regulatory standards. Organizations like the U.S. Food and Drug Administration and the World Health Organization enforce stringent GMP guidelines to ensure drug safety and quality.
Companies such as Ipca Laboratories Limited play a vital role in supplying high-quality APIs globally, making them key employers for experienced pharma professionals.

