Ipca Laboratories Limited is a globally recognized pharmaceutical company known for high-quality API manufacturing and finished dosage formulations.
With a strong presence in regulated markets like the US and Europe, the company follows strict compliance standards such as WHO-GMP, MHRA, and USFDA, making it a preferred employer in the pharma industry.
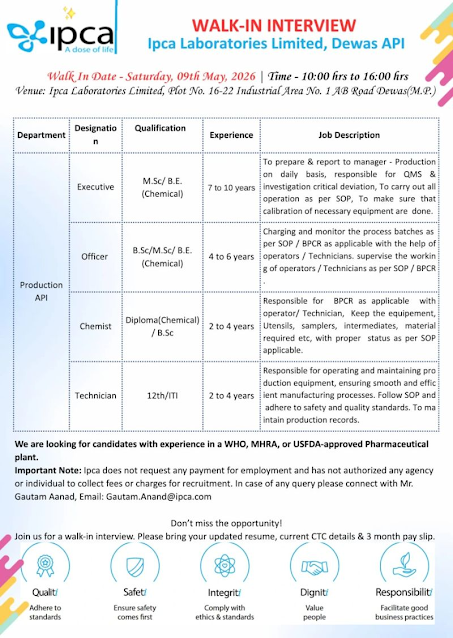
Ipca Laboratories Walk-In Interview 2026 – Highlights
- Company Name: Ipca Laboratories Limited
- Department: Production (API)
- Experience Required: 2 to 10 Years
- Location: Dewas
Job Openings Details
1) Executive – API Production
- Experience: 7–10 Years
- Qualification: M.Sc / B.E. (Chemical)
Key Responsibilities:
- Daily production reporting
- Handling QMS & critical deviation investigations
- Ensuring SOP compliance
- Equipment calibration monitoring
2) Officer – API Production
- Experience: 4–6 Years
- Qualification: B.Sc / M.Sc / B.E. (Chemical)
Key Responsibilities:
- Batch charging & monitoring
- Supervising operators & technicians
- Following SOP / BPCR processes
3) Chemist – API Production
- Experience: 2–4 Years
- Qualification: Diploma (Chemical) / B.Sc
Key Responsibilities:
- Managing BPCR documentation
- Maintaining equipment and materials
- Ensuring SOP compliance
4) Technician – API Production
- Experience: 2–4 Years
- Qualification: 12th / ITI
Key Responsibilities:
- Operating production equipment
- Maintaining manufacturing efficiency
- Recording production data
- Following safety and quality standards
Walk-In Interview Details
- 📅 Date: 9 May 2026 (Saturday)
- ⏰ Time: 10:00 AM – 4:00 PM
- 📌 Location: Dewas
Venue:
Ipca Laboratories Limited
Plot No. 16–22, Industrial Area No. 1
AB Road, Dewas (M.P.)
Who Can Apply
✔ Candidates with 2–10 years of API production experience
✔ Experience in regulated plants like WHO, MHRA, or USFDA
✔ Professionals with strong knowledge of SOP-based manufacturing
✔ Candidates from Chemical / Pharma / Science backgrounds
Career Growth at Ipca Laboratories
Working at Ipca Laboratories Limited offers:
✨ Exposure to global regulatory standards
✨ Hands-on experience in API manufacturing
✨ Strong knowledge of QMS & compliance
✨ Long-term career growth in pharma production
✨ Opportunities in international markets
Why Choose Ipca Laboratories?
- Global pharmaceutical presence
- Strong compliance with WHO-GMP & USFDA
- Advanced API manufacturing facilities
- Excellent learning and career development
- Trusted brand in pharma industry
Important Notes
✔ No recruitment fees charged by the company
✔ No third-party hiring agencies involved
✔ Carry all required documents for verification
✔ Ensure eligibility before attending
FAQs
Q1. What is the job location?
👉 Dewas, Madhya Pradesh
Q2. What experience is required?
👉 2–10 years in API production and regulated pharma plants
Q3. Is there any application fee?
👉 No, the recruitment process is completely free

