Looking to grow your career in sterile pharmaceutical manufacturing? The latest Gufic Biosciences Hiring 2026 drive brings exciting opportunities for experienced professionals in multiple departments at its injectable facility in Navsari.
Gufic Biosciences Limited is inviting applications for Officer / Sr. Officer roles in QA, QC (Microbiology), Production, Maintenance, HVAC, and Warehouse.
About Gufic Biosciences Limited
Founded in 1970, Gufic Biosciences Limited is a leading name in Lyophilized Injectable manufacturing with over 50 years of expertise.
Global Presence & Approvals
The company holds multiple international regulatory approvals, including:
- WHO-GMP
- EU-GMP
- Canada
- Russia
- ANVISA
- Other global regulatory authorities
Its modern facility in Navsari is designed to meet strict global compliance standards.
Job Overview
- Company: Gufic Biosciences Limited
- Location: Navsari, Gujarat
- Designation: Officer / Sr. Officer
- Experience Required: 2 to 8 years
- Industry: Injectable / Sterile Manufacturing
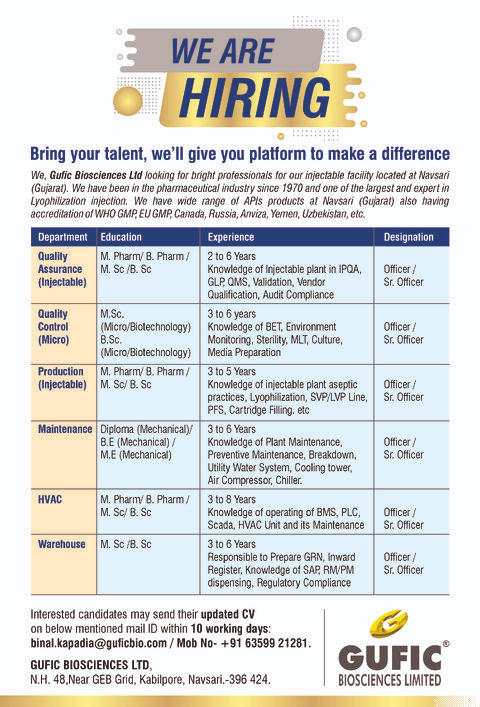
Current Openings
Quality Assurance (Injectable)
- Qualification: M.Pharm / B.Pharm / M.Sc / B.Sc
- Experience: 2–6 years
- Skills: IPQA, QMS, GLP, Validation, Audit Compliance
Quality Control (Microbiology)
- Qualification: M.Sc / B.Sc (Microbiology / Biotechnology)
- Experience: 3–6 years
- Skills: Sterility Testing, BET, Environmental Monitoring, MLT
Production (Injectable)
- Qualification: M.Pharm / B.Pharm / M.Sc / B.Sc
- Experience: 3–5 years
- Skills: Aseptic Practices, Lyophilization, SVP/LVP, PFS
Maintenance (Mechanical)
- Qualification: Diploma / B.E / M.E (Mechanical)
- Experience: 3–6 years
- Skills: Plant Maintenance, Utility Systems, Chillers, Compressors
HVAC
- Qualification: Pharma/Science Background
- Experience: 3–8 years
- Skills: BMS, PLC, SCADA, HVAC Operations
Warehouse
- Qualification: M.Sc / B.Sc
- Experience: 3–6 years
- Skills: SAP, GRN, RM/PM Handling, Compliance
Required Skills & Qualifications
Candidates applying under Gufic Biosciences Hiring should have:
- Experience in Injectable / Sterile / Lyophilization plants
- Strong knowledge of cGMP and GLP guidelines
- Hands-on technical expertise in respective domains
- Good documentation and communication skills
Verified Post
Verification: To confirm the legitimacy of this posting, you can view the original announcement on the Gufic LinkedIn page.
How to Apply
Email Application
Send your updated CV to: binal.kapadia@guficbio.com
📱 Contact: +91 63599 21281
👉 Important: Mention your Department & Position in the subject line.
Plant Address
Gufic Biosciences Ltd
N.H. 48, Near GEB Grid, Kabilpore,
Navsari – 396424, Gujarat
Why Choose Gufic Biosciences?
Joining Gufic Biosciences Limited offers:
- Exposure to advanced Lyophilization technology
- Work in a globally approved injectable facility
- Strong career growth in sterile manufacturing
- Hands-on experience with international compliance systems
This makes it one of the best opportunities for professionals aiming to excel in injectable pharma careers.
FAQs
Q1: Which departments are hiring?
👉 QA, QC (Micro), Production, Maintenance, HVAC, and Warehouse
Q2: What experience is required?
👉 2–8 years depending on the role
Q3: Is injectable experience necessary?
👉 Yes, sterile/lyophilization experience is highly preferred
Q4: How can I apply?
👉 Send your CV via email or contact the provided number

