sterile pharma jobs in Gujarat? The latest Innoxel Lifesciences Walk-In Interview 2026 offers excellent career opportunities for experienced professionals in Quality Control, Production, Quality Assurance, Validation, Engineering, and more.
Innoxel Lifesciences Pvt. Ltd. is conducting a mega walk-in drive on 22 March 2026 at its Vadodara facility, targeting candidates with USFDA sterile manufacturing experience.
About Innoxel Lifesciences
Innoxel Lifesciences Pvt. Ltd. is a fast-growing pharmaceutical company focused on:
-
Sterile injectables manufacturing
-
Liquid oral formulations
-
Compliance with USFDA and global regulatory standards
Located in Vadodara, the company provides:
-
Advanced sterile manufacturing infrastructure
-
Strong focus on quality and compliance
-
Career growth in regulated pharmaceutical environments
Walk-In Interview Details
| Details | Information |
|---|---|
| Company | Innoxel Lifesciences Pvt. Ltd. |
| Date | 22 March 2026 |
| Time | 10:00 AM – 04:00 PM |
| Location | Vadodara, Gujarat |
📍 Venue:
Survey No. 534/1, Village Kotambi, Near VCA Stadium, Tal. Waghodia, Vadodara, Gujarat
📧 Email Application:
hr1@innoxells.com
Job Openings by Department
Quality Control (QC)
-
Role: Chemist / Analyst (AMV, RM/PM)
-
Experience: 1–5 Years
-
Qualification: B.Sc / M.Sc / B.Pharm / M.Pharm
-
Skills: Analytical testing, method validation
Production – Sterile / Liquid Oral
-
Role: Operator / Supervisor
-
Experience: 1–8 Years
-
Qualification: ITI / Diploma / B.Pharm / M.Pharm
-
Skills:
-
Aseptic filling
-
Autoclave, vial washing, tunnel
-
SKID operations
-
QC Microbiology
-
Role: Microbiologist
-
Experience: 1–5 Years
-
Qualification: B.Sc / M.Sc Microbiology
-
Skills:
-
Environmental monitoring (EM)
-
Sterility testing
-
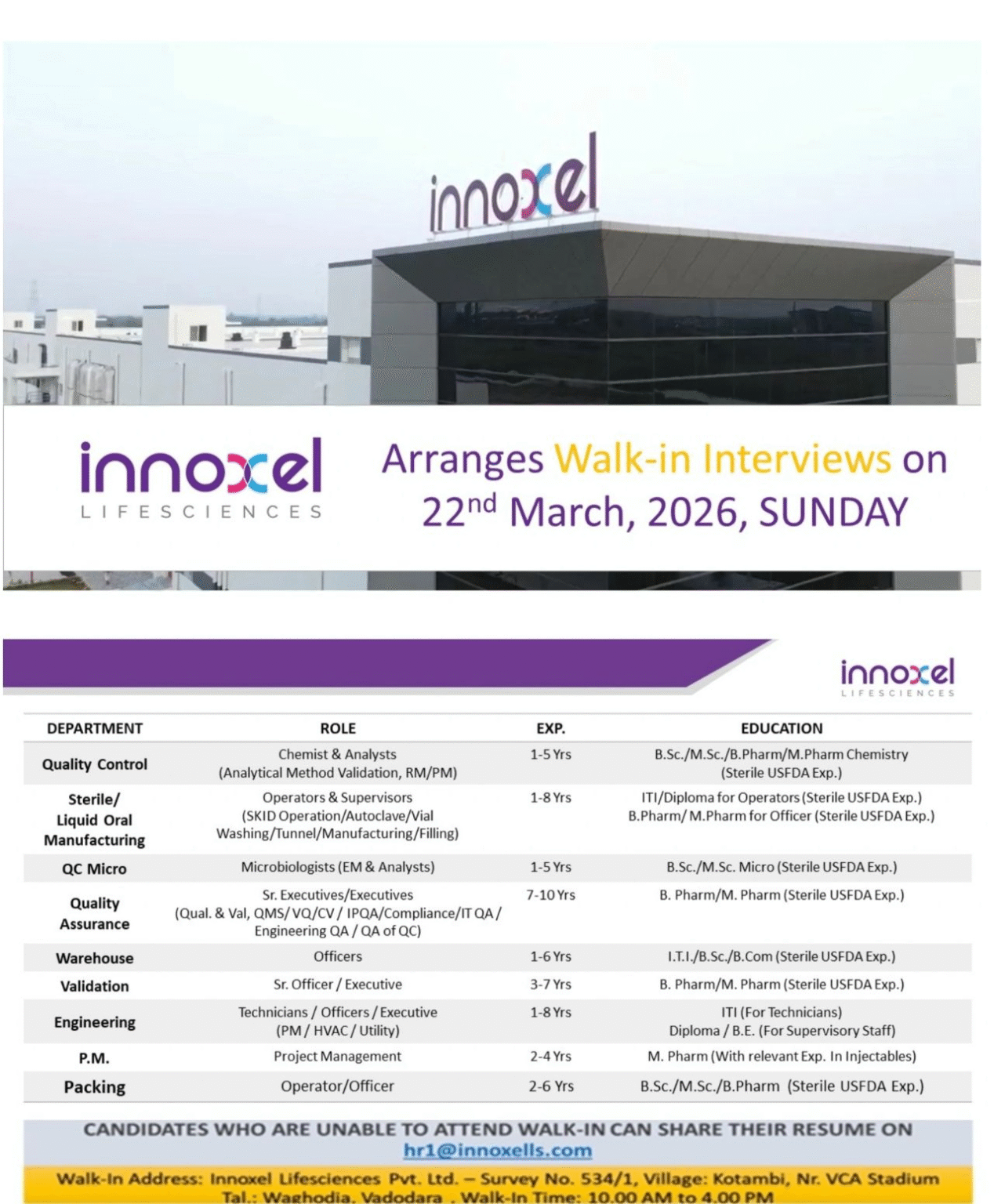
Quality Assurance (QA)
-
Role: Executive / Sr. Executive
-
Experience: 7–10 Years
-
Qualification: B.Pharm / M.Pharm
-
Skills:
-
QMS, IPQA, validation
-
Compliance & audit readiness
-
Validation
-
Role: Sr. Officer / Executive
-
Experience: 3–7 Years
-
Qualification: B.Pharm / M.Pharm
Engineering
-
Role: Technician / Officer / Executive
-
Experience: 1–8 Years
-
Qualification: ITI / Diploma / B.E
-
Skills: HVAC, utilities, plant maintenance
Warehouse
-
Role: Officer
-
Experience: 1–6 Years
-
Qualification: ITI / B.Sc / B.Com
Packing
-
Role: Operator / Officer
-
Experience: 2–6 Years
-
Qualification: B.Sc / B.Pharm
Key Skills Required
Candidates applying for Innoxel Lifesciences jobs should have:
-
Experience in USFDA-regulated sterile environments
-
Knowledge of:
-
Aseptic processing
-
Analytical instruments
-
GMP documentation
-
-
Expertise in:
-
Environmental monitoring
-
Validation protocols
-
Compliance and audits
-
Who Can Apply?
-
Professionals with 1–10 years of experience (role-dependent)
-
Candidates with pharma/sterile manufacturing background
-
Freshers may check with HR for trainee roles
Why Join Innoxel Lifesciences?
Joining Innoxel Lifesciences Pvt. Ltd. offers:
-
Work in a USFDA-compliant sterile facility
-
Hands-on experience with advanced aseptic technologies
-
Career growth in QA, QC, and production domains
-
Exposure to global pharmaceutical standards
-
Stable and growth-oriented work environment
How to Apply
Walk-In Process
📅 Date: 22 March 2026
⏰ Time: 10:00 AM – 04:00 PM
Carry:
-
Updated resume
-
Educational documents
-
Experience certificates
-
Salary documents
Email Option
📧 Send your CV to: hr1@innoxells.com
Industry Background – Sterile Pharma Growth in Gujarat
Gujarat is a leading hub for:
-
Sterile injectables manufacturing
-
USFDA-approved pharma facilities
-
Export-driven pharmaceutical production
Cities like Vadodara are witnessing rapid growth in regulated pharma jobs, especially in QA, QC, and sterile production.
Authoritative Sources
For pharmaceutical compliance and sterile manufacturing standards:
-
Central Drugs Standard Control Organization (CDSCO)
-
United States Food and Drug Administration
-
World Health Organization – GMP Guidelines
-
Indian Pharmaceutical Alliance
These authorities define the standards followed by companies like Innoxel Lifesciences.
FAQs – Innoxel Lifesciences Walk-In Interview 2026
1. What experience is required?
Candidates should have 1–8 years of sterile pharma experience, depending on the role.
2. Are freshers eligible?
This drive mainly targets experienced candidates, but freshers can check with HR for trainee roles.
3. Where is the walk-in venue?
At Village Kotambi, Waghodia, Vadodara, Gujarat.
4. What roles are available?
Roles are open in:
-
QC
-
QA
-
Production
-
Validation
-
Engineering
-
Warehouse & Packing
5. What growth opportunities are available?
Strong career progression in QA/QC and sterile manufacturing, with exposure to global regulatory systems

